Abstract
Myeloid cells are the most evolutionarily ancient aspect of a specialized immune system and the cornerstone of innate immunity in vertebrates. Innate immunity serves at the front line of host defense playing essential roles in directly clearing infection while also activating adaptive immune cell populations. Even with the importance in its roles, the myeloid system has often been regarded as the 'dumb brute' side of cell-based immunity with limited specificity and variability of responses. Heterogeneity in innate immune cells is increasingly recognized but still modest compared to other cell types and constrained by limited investigative tools.
Monocytes have traditionally been categorized into two subsets: classical and non-classical. Classical monocytes are thought to readily convert to non-classical monocytes with exogenous cues. Studies on bulk monocyte populations have shown that monocytes acquire functional features dependent on physiological needs supporting a prevailing model that monocytes are highly plastic.
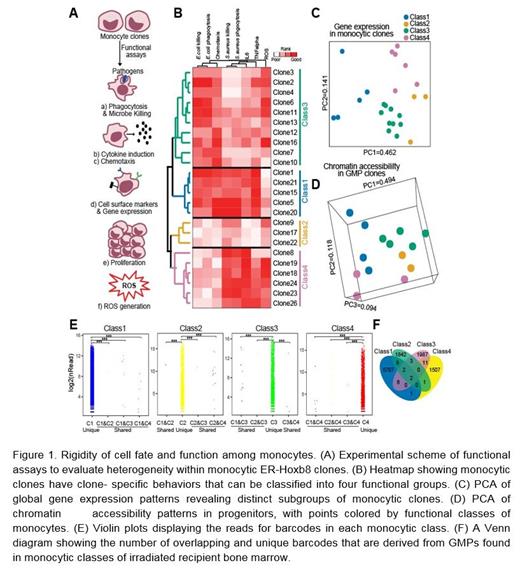
We adapted a system for inducible clonal expansion of mouse granulocyte-monocyte progenitor (GMP) capable of differentiating into mature myeloid cells to address at a clonal level the issues of monocyte heterogeneity, plasticity, and whether changes in monocyte functional groups are due to induction or selection, Using inducible HoxB8-ER, large numbers of primary self-renewing GMP can undergo progressive maturation to fully functional granulocytes or monocytes upon removal of estrogen. The resulting GMP clones could then be characterized molecularly and immunophenotypically in correlation with the specialized functions of their descendent monocytes (Figure 1A). Four GMP differentiation trajectories that yield functionally distinct monocytes were defined (Figures 1B and 1C). Notably the functional monocyte groups were evident at the GMP level based on unsupervised clustering of chromatin configuration data (Figure 1D). GMP bore epigenetic scripting of the potential of their descendent monocytes and this fate had little flexibility once differentiation had begun. Testing cells under in vitro and in vivo homeostasis and stress conditions revealed that the cells maintain their differentiation path and do not transition from one state to another (Figures 1E and 1F). These data imply a model whereby monocyte stimulus-specific selection may occur and may contribute to an innate immune memory that resembles adaptive immune memory.
Sykes: Keros Therapeutics: Consultancy; SAFI Biosolutions: Consultancy, Current equity holder in publicly-traded company; Clear Creek Bio: Current equity holder in publicly-traded company. Scadden: Magenta Therapeutics: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Clear Creek Bio: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; LifeVaultBio: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Editas Medicines: Membership on an entity's Board of Directors or advisory committees; Fate Therapeutics: Current holder of individual stocks in a privately-held company; Clear Creek Bio: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Dainippon Sumitomo Pharma: Other: Sponsored research; FOG Pharma:: Consultancy; Garuda Therapeutics: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; VCanBio: Consultancy; Inzen Therapeutics: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal